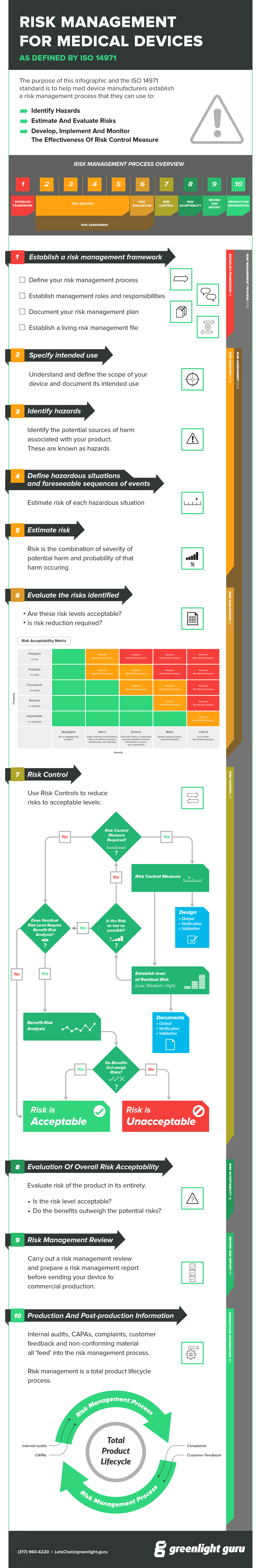 
The main purpose of FMEA is to identify potential design problems early on in the process so that they can be eliminated or reduced before they can negatively affect the safety or effectiveness of the final device. FMEA is a bottom-up method intended to evaluate the effects of defects at the component level and determine appropriate design changes. Failure mode effects analysis is used during the design phase of the device life cycle. It is this risk/benefit ratio that medical device manufacturers need to work with by keeping the associated device risks as low as possible.Īn effective way to manage this process is to employ a risk analysis using such tools as Failure Mode Effects Analysis (FMEA) or Fault Tree Analysis (FTA). While we cannot change the fact that every device from simple to the most complex has inherent risks, we must identify such risks, study and mitigate them wherever possible, and then weigh them in against the device benefits. We all know examples of sad stories of patients who were overdosed on radiotherapy devices, injured by electric shocks, over-infused on the infusion pump, and so many other tragic cases of death and patient injuries. 
Risk Analysis for the Medical Device Industryīeing in the medical device industry means understanding and properly applying the risk analysis principles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
